Image source: SA Health
This article is meant to raise awareness of the meningococcal B vaccine. It is not meant to replace discussion with, or the advice of your own family GP.
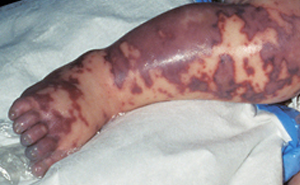
The “M” word – meningococcal, scares the life out of any parent with a sick child. Although rare (there are only around 200 cases per year1,2), everyone has heard of cases resulting in deafness, brain damage, loss of limbs and death. Unfortunately, it can be a difficult disease to diagnose as it presents with common symptoms such as fever, irritability, headache, neck stiffness, muscles aches and vomiting. The characteristic rash, which looks like little red/purple bruises is not always present2. The highest incidence of Meningococcal disease is in infants under two years with another peak in adolescence1,2.
Why meningococcal B?
Meningococcal disease is caused by various different strains of the bacteria, “Neisseria meningitidis”. In Australia strains B and C are the most common3. Currently children are immunised against the C strain at 12 months. Although potentially serious this is not the most common cause of meningococcal disease in Australia. The cases you hear in the news of children losing limbs and lives are usually meningococcal B. About 85% of meningococcal in recent years has been caused by B strain2.
About the meningococcal B vaccine (“Bexsero”)
- Available in Australia since March 2014. It is not on the immunisation schedule, so is only available privately (meaning it is expensive – about $120 per injection)
- Registered for use in babies from 2 months of age5.
- Babies <6 months require a total of 4 injections (3 injections 2 months apart and a booster at 12m or 2 months after the last injection whichever is later)
- Babies 6-11 months require 3 injections (2 injections 2 months apart and a booster at 12months or 2 months after the last injection, whichever is later).
- Babies >12 months require 2 injections (2 injections 2 months apart).
- Covers about 76% of Meningococcal B in Australia 1,2 (so is not 100% effective)
- At this point, duration of protection against the disease is unknown (so at some point after immunisation children may not be protected anymore)
- Although lab studies show a good immune response generated by the immunisation, exactly how effective the vaccine is in preventing the disease in real life is not known2 (It is hardly ethical to “give” children Meningococcal disease to see how well the vaccine works!)
- It has been used since 2013 in the UK. It was added to the UK immunisation schedule in September 2015.
How safe is it and what are the side effects?
- It cannot cause Meningococcal disease.
- Clinical trials have been conducted in over 8000 people (5000 babies) and it has been deemed safe4. More than 500 000 doses have now been given worldwide.
- Fever is the most common side effect. In fact, it is recommended paracetamol be given half an hour before the injection1,2.
- Note that it can be given with routine immunisations at 2, 4, 6 months however this is associated with more fevers and injection site reactions 2,5.
- Other side effects include injection site redness or pain, sleepiness, irritability and appetite changes
- Development of fever may (rarely) lead to febrile seizures. Although scary to witness, 3% of healthy children will experience one between six months and six years regardless of immunisation. Rarely are there any long-term consequences.
So why isn’t it on the National Immunisation Program?
- The committee that make recommendations to the government on immunisations has not recommended it be included on our schedule but NOT due to safety concerns1.
- The issue is more one of cost-effectiveness (its an expensive vaccine and a very rare disease) and uncertainty regarding duration of protection offered by the vaccine/uncertainly regarding exactly how well it works in real life1.
So, my thoughts (just my opinion…)
I did vaccine my first baby. I chose to do it separately from his routine immunisations because of increased side effects when administered together. I was happy with the safety profile and decided that even if he was partially covered and only for a couple of years it was worth doing since he is most at risk in his first two years of life. Although the disease is rare, the consequences are commonly serious and/or fatal so I felt the benefits outweighed the cost. Because of the uncertainties and the high cost, I wouldn’t push it on patients but I think it is important to be aware of the benefits/limitations so together with your GP you can make the decision.
I have now immunised both of my children. While my son was irritable post his doses, my daughter was completely fine apart from a big red mark on her leg (which settled with time and didn’t bother her). What I didn’t think about the first time around was to check with my private health insurance. My policy gave me about $80 back for each of my daughter’s doses, making it much more affordable!
Information from:
- The Australian Government Pharmaceutical Benefits Scheme website (http://www.pbs.gov.au/info/industry/listing/elements/pbac-meetings/psd/2013-11/meningococcal-vaccine)
- National Centre for Immunisation Research & Surveillance (NCIRS) (Meningococcal vaccines for Australians fact sheet_1 July 2020_Final_0.pdf (ncirs.org.au))
- Immunise Australia website (http://www.immunise.health.gov.au/internet/immunise/publishing.nsf/Content/immunise-meningococcal)
- Meningitis Research Foundation (www.meningitis.org/menb-vaccine













